FEBRUARY 19, 2026, NEW YORK – In a groundbreaking development poised to reshape cancer treatment, researchers have engineered a novel type of chimeric antigen-receptor (CAR) T cell that can be remotely controlled, potentially mitigating the severe side effects associated with current immunotherapy. This innovation, detailed in the latest issue of Nature Chemical Biology, promises a safer and more effective approach to treating various forms of cancer.
CAR-T cells, a promising tool in the fight against cancer, have already revolutionized the treatment of blood cancers. However, their effectiveness against solid tumors has been limited, and they often pose risks of damaging healthy cells or triggering severe immune responses. To address these challenges, a team led by Melita Irving and Greta Maria Paola Giordano Attianese from Ludwig Lausanne, in collaboration with Leo Scheller and Bruno Correia from the École Polytechnique Fédérale de Lausanne (EPFL), has developed a CAR-T cell that can be switched off on demand using a cancer drug already in clinical use.
Innovative Design for Enhanced Control
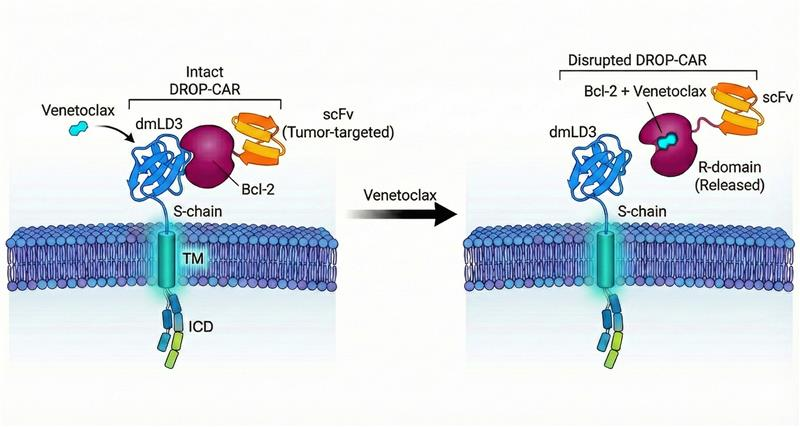
The researchers introduced a new design known as “drug-regulated off-switch PPI CAR” (DROP-CAR), which allows for reversible control of CAR-T cell activation. This system employs venetoclax, a drug currently used in cancer therapy, to disengage the CAR-T cells from their cancerous targets without destroying the cells themselves. This design not only enhances safety but also broadens the potential application of CAR-T therapy.
“Our work introduces a simple and clinically realistic way to reversibly dial down CAR-T cell activation,” said Irving. “The remote control doesn’t trigger the self-destruction of the CAR-T cells but simply prompts them to disengage and fall off from their cancerous targets.”
The Mechanics Behind DROP-CAR
Traditional CAR-T cells feature a protein receptor that detects cancer cells, triggering a cytotoxic response. The new DROP-CAR design places a switch on the outside of the cell, using a protein-protein interaction to hold the CAR together. When venetoclax is introduced, this interaction is disrupted, effectively turning off the CAR-T cells. Once the drug is withdrawn, the CAR reassembles, allowing the cells to resume their cancer-fighting function.
“Unlike previous controllable CAR designs, our system uses only human protein components and a clinically approved, non-immunosuppressive drug to directly disrupt tumor cell binding,” explained Giordano Attianese.
Addressing T Cell Exhaustion
A significant advantage of the DROP-CAR system is its potential to combat T cell “exhaustion,” a common issue in immunotherapy where continuous stimulation renders T cells ineffective. By allowing CAR-T cells periods of rest, the system can reverse genomic alterations that lead to exhaustion, thereby boosting their efficacy. This approach aligns with previous studies suggesting that intermittent rest can enhance T cell performance.
The ability to modulate CAR-T cell activity without sacrificing the cells themselves represents a major advancement in cancer treatment, offering a more controlled and sustained therapeutic option.
Pathway to Clinical Application
With the controlling drug already approved for cancer therapy, the DROP-CAR system is uniquely positioned for clinical evaluation. The researchers are optimistic about its potential to transform CAR-T therapy, making it accessible to a broader range of patients and cancer types.
Supported by organizations such as the Swiss National Science Foundation and the European Research Council, this research marks a significant step forward in the field of cancer immunotherapy. As the scientific community continues to explore the potential of engineered immune cells, innovations like DROP-CAR could pave the way for more effective and safer cancer treatments.
The announcement comes as an encouraging development in the ongoing battle against cancer, offering hope for improved outcomes and reduced side effects for patients worldwide. As clinical trials progress, the medical community eagerly anticipates the potential impacts of this innovative therapy.